HOOKVAC
Developing and testing a novel, low-cost, effective HOOKworm VACcine to control human hookworm infection in endemic countries

Objective
HOOKVAC will address four main objectives:
1. Establish safety and immunogenicity of the vaccine candidate in an endemic population;
2. Improve the manufacturing process;
3. Provide clinical proof of concept; and
4. Improve accessibility of the vaccine in endemic areas.
Description
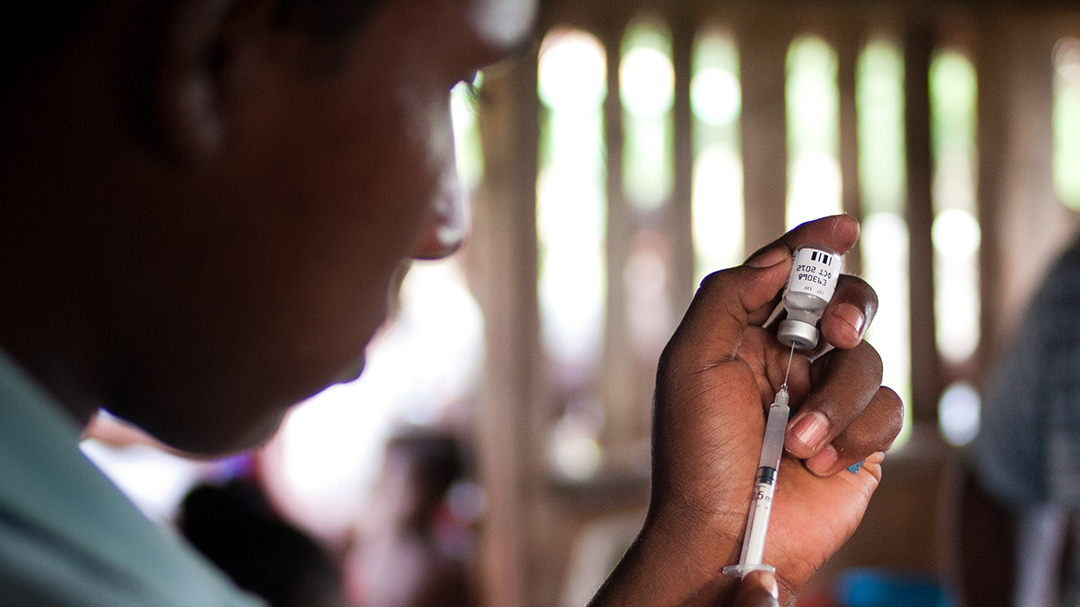
HOOKVAC studies a candidate vaccine against hookworm infection, which ranks number one in terms of years lost from disability from a neglected infectious disease. It addresses four main objectives: 1. Establish safety and immunogenicity of the vaccine candidate in an endemic population; 2. Improve the manufacturing process; 3. Provide clinical proof of concept; and 4. Improve accessibility of the vaccine in endemic areas (sub-Saharan Africa, Southeast Asia and Latin America). HOOKVAC is developing the first and only vaccine for human hookworm infection. A bivalent, low-cost vaccine candidate is being clinically tested for the first time in an African disease endemic population. The study is being conducted in Gabon.
Partners
AMC
Albert B. Sabin Vaccine Institute (USA)
Eberhard Karls Universitaet Tuebingen (Germany)
Centre de Recherches Medicales de Lamberéné (Gabon)
Q-Biologicals (Belgium)
Pharmidex Pharmaceutical Services (United Kingdom)
Academisch Ziekenhuis Leiden – Leids Universitair Medisch Centrum (Netherlands)
Baylor College of Medicine (USA)
George Washington University (USA)
Funders
European Union’s Seventh Framework Programme
Countries
Netherlands
Germany
Belgium
Gabon
United Kingdom
United States
Links
New York Times Parasites: Hookworm Vaccine Will Be Tried in Africa
The Sabin Vaccine Institute Phase 1 Clinical Trial of Human Hookworm Vaccine Successfully Completed
